Hund’s Coupling Cases for Diatomic Molecules
In diatomic molecules, electronic angular momenta can couple to the internuclear axis in different ways depending on the relative strengths of spin–orbit coupling and rotational motion. These limiting behaviors are described by Hund’s coupling cases. The most common cases for light and moderately heavy diatomic molecules are Hund’s case (a) and Hund’s case (b).
Hund’s cases are idealized limits: real molecules may lie between them, but the cases provide a powerful framework for constructing term symbols, predicting level structure, and understanding spectra.
Hund’s Case (a): Strong Spin–Orbit Coupling
Hund’s case (a) applies when spin–orbit coupling is strong compared to rotational energy. In this limit, both the electronic orbital angular momentum and the electron spin are strongly tied to the internuclear axis.
- The projection of electronic orbital angular momentum on the molecular axis is \(\Lambda = 0,1,2,\ldots \Rightarrow \Sigma,\Pi,\Delta,\ldots\).
- The projection of electronic spin on the molecular axis is \(\Sigma = -S, -S+1, \ldots, +S\).
- These combine to give \(\Omega = \Lambda + \Sigma\), the projection of the total electronic angular momentum on the axis.
- Molecular rotation then adds angular momentum to form \(J = \Omega, \Omega+1, \Omega+2, \ldots\).
In case (a), \(\Omega\) is a good quantum number, and molecular term symbols are written as \({}^{2S+1}\Lambda_{\Omega}\) (with additional parity and symmetry labels added later).
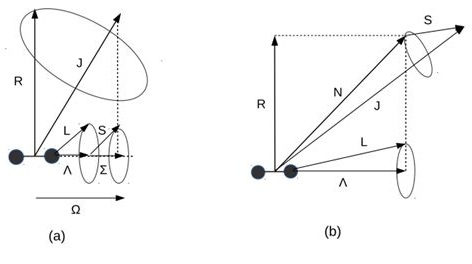
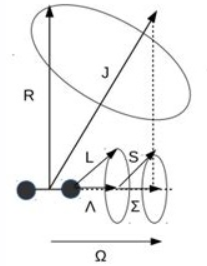
Hund’s coupling case (a): electronic orbital and spin angular momenta (Λ, Σ) couple strongly to the molecular axis, giving Ω = Λ + Σ; rotation R then couples to form the total J.
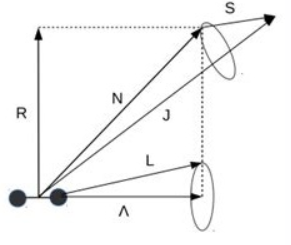
Hund’s Case (b): Weak Spin–Orbit Coupling
Hund’s case (b) applies when spin–orbit coupling is weak compared to rotational energy. In this limit, the electronic orbital angular momentum remains coupled to the internuclear axis, but the electron spin does not.
- The projection of orbital angular momentum, \(\Lambda\), remains a good quantum number.
- The electron spin \(S\) is only weakly coupled and instead interacts primarily with molecular rotation.
- Molecular rotation is described by \(\mathbf{N}\), the total angular momentum excluding spin.
- The total angular momentum is then \(\mathbf{J} = \mathbf{N} + \mathbf{S}\).
In case (b), \(\Omega\) is not a good quantum number. Levels are instead labeled by \(N\) and \(J = N+S,\,N+S-1,\ldots,|N-S|\).
Summary: Hund’s case (a) emphasizes coupling to the molecular axis (\(\Lambda, \Sigma, \Omega\) good), while Hund’s case (b) emphasizes rotational coupling (\(\Lambda, N, J\) good). Identifying the appropriate case is the first step in interpreting diatomic molecular spectra.