Solving the radial Schrödinger equation for the hydrogen atom reveals an important feature of quantum mechanics: only specific energy values lead to physically acceptable wavefunctions. These allowed energies are the energy eigenvalues of the hydrogen atom.
Normalizable radial wavefunctions
The radial wavefunction \(R(r)\) must be normalizable, meaning that the total probability of finding the electron somewhere in space is finite:
\[ \int_0^\infty |R(r)|^2 r^2\,dr < \infty. \]
For most values of the energy, the radial solutions diverge either as \(r \to 0\) or as \(r \to \infty\), making them unphysical. Only a discrete set of energies produces radial wavefunctions that remain finite everywhere and decay properly at large \(r\).
These discrete energies are labeled by the principal quantum number \(n = 1,2,3,\ldots\).
Hydrogen atom energy eigenvalues
The allowed energy levels of the hydrogen atom are
\[ E_n = -\frac{\mu e^4}{2(4\pi\varepsilon_0)^2\hbar^2} \frac{1}{n^2} = -\frac{hc\,R_M}{n^2}, \]
where \(R_M\) is the mass-corrected Rydberg constant. (See the Rydberg Constant for more details.)
It is important to note that the energy depends only on \(n\). It does not depend on the angular momentum quantum number \(l\) or the magnetic quantum number \(m_l\).
As a result, all states with the same \(n\) but different \(l\) and \(m_l\) are degenerate in energy.
Spacing of energy levels
\(n\) increases. At low \(n\), the energy levels are widely separated, while at high \(n\) they become increasingly close together.
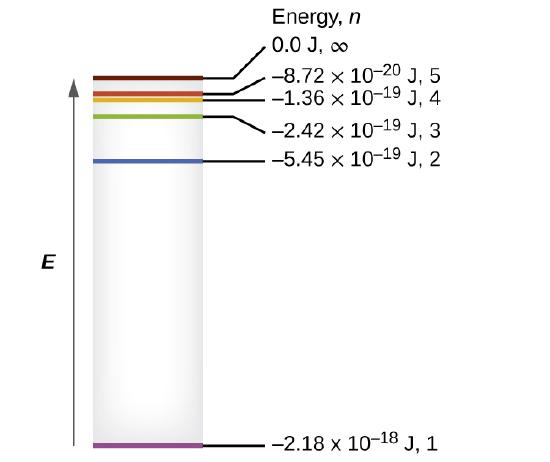
In the limit \(n \to \infty\), the energy approaches zero from below:
\[ \lim_{n\to\infty} E_n = 0. \]
This limit corresponds to the ionization energy of the hydrogen atom—the point at which the electron is no longer bound to the nucleus.
Connection to the Rydberg equation
Spectral lines arise from transitions between two allowed energy levels, \(n_i\) and \(n_f\). The energy difference is
\[ \Delta E = E_{n_f} - E_{n_i} = -hcR_M\left(\frac{1}{n_f^2}-\frac{1}{n_i^2}\right). \]
Dividing by \(hc\) gives the wavenumber of the emitted or absorbed radiation:
\[ \tilde{\nu} = R_M\left(\frac{1}{n_f^2}-\frac{1}{n_i^2}\right), \]
which is the Rydberg equation.
Big idea: requiring the hydrogen atom wavefunction to be normalizable leads to discrete energy levels that depend only on \(n\), whose differences explain the observed atomic line spectra.
