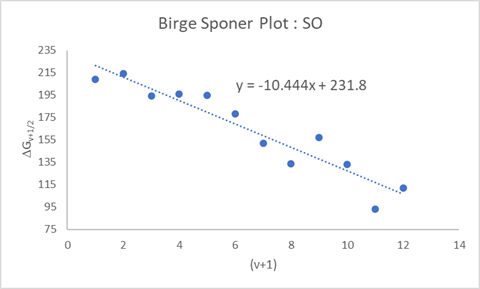
The Birge–Sponer extrapolation is a graphical method used to estimate the dissociation energy of a diatomic molecule from vibrational spectroscopy data. It exploits the systematic decrease in vibrational level spacing caused by anharmonicity.
The key idea is that vibrational level spacings, \(\Delta G_{v+1/2}\), decrease smoothly as the vibrational quantum number increases and eventually approach zero at dissociation.
Step 1: Use measured vibrational spacings
From a spectrum (typically UV-visible, but could also be infrared), one can determine the spacings between successive vibrational levels within a single electronic state of a molecule:
\[ \Delta G_{v+1/2} = G_{v+1} - G_v. \]
For an anharmonic oscillator, these spacings are well approximated by
\[ \Delta G_{v+1/2} = \omega_e - 2\omega_e x_e (v+1). \]
This expression predicts a linear decrease of \(\Delta G_{v+1/2}\) with increasing \((v+1)\).
Step 2: Birge–Sponer plot
In the Birge–Sponer method, the measured spacings \(\Delta G_{v+1/2}\) are plotted against \((v+1)\).
- The vertical intercept of the line gives \(\omega_e\).
- The slope of the line is \(-2\omega_e x_e\).
From a linear fit to the data, both \(\omega_e\) and \(\omega_e x_e\) can be extracted directly.
Step 3: Determining the highest bound level
Dissociation occurs when the spacing between vibrational levels goes to zero:
\[ \Delta G_{v+1/2} = 0. \]
Extrapolating the fitted line to zero spacing yields the maximum vibrational quantum number, \(v_{\max}\), corresponding to the highest bound vibrational level.
Step 4: Determining the dissociation energy \(D_0\)
The dissociation energy from the lowest vibrational level, \(D_0\), is obtained by summing (or integrating) all vibrational spacings up to dissociation:
\[ D_0 = \sum_{v=0}^{v_{\max}} \Delta G_{v+1/2}. \]
Graphically, this corresponds to the area under the Birge–Sponer plot from \((v+1)=0\) to \((v+1)=v_{\max}+1\).
In practice, the area is evaluated using the fitted straight line, providing an estimate of \(D_0\) in spectroscopic units (\(\text{cm}^{-1}\)).
Big idea: the Birge–Sponer extrapolation uses anharmonic vibrational spacings to estimate molecular dissociation energies by extrapolating to the point where vibrational binding disappears.