The Free Electron Model
an application of the Particle in a Box Problem
Worked Example: 1,3-Butadiene (Kuhn Free Electron Model)
We will use Kuhn’s free electron model to estimate the HOMO–LUMO gap and the absorption wavelength for 1,3-butadiene (\( \mathrm{C_4H_6} \)). The key steps are: (1) estimate the box length \(L\), (2) count \( \pi \) electrons, (3) fill energy levels, (4) compute the HOMO–LUMO energy gap, and (5) convert to a wavelength.
Step 1: Estimate the box length, \(L\)
Butadiene has 4 conjugated carbon atoms, so its conjugated backbone contains \(N=4\) carbons. A simple (common) approximation is: take an average C–C spacing of about \(1.40~\text{Å}\) (which is roughly the C-C distance in benzene), and add “half a bond” at each end. This makes the box length approximately
\[ L \approx N(1.40~\text{Å}) = 4(1.40~\text{Å}) = 5.6~\text{Å} = 5.6\times 10^{-10}~\text{m}. \]
(This end-correction is a simple way to reflect that the electron density does not abruptly stop exactly at the terminal carbons.)
Step 2: Count the \( \pi \) electrons
Each carbon atom in the cunjugated backbone contributes one \( \pi \) electron, so butadiene has
\[ N_{\pi} = 4~\pi\text{ electrons}. \]
Step 3: Fill the particle-in-a-box levels
The particle-in-a-box energies are
\[ E_n = \frac{n^2h^2}{8mL^2}, \qquad n=1,2,3,\dots \]
Each energy level holds 2 electrons (with paired spins). With 4 electrons:
- \(n=1\) holds 2 electrons (filled)
- \(n=2\) holds 2 electrons (filled) → this is the HOMO
- \(n=3\) is the next level → this is the LUMO
So for butadiene: \(n_{\text{HOMO}}=2\) and \(n_{\text{LUMO}}=3\).
Step 4: Compute the HOMO–LUMO energy gap
The predicted gap is \( \Delta E = E_{3}-E_{2} \):
\[ \Delta E = \frac{h^2}{8mL^2}\left(3^2-2^2\right) = \frac{5h^2}{8mL^2}. \]
Using \(h=6.626\times10^{-34}~\text{J·s}\), \(m=m_e=9.11\times10^{-31}~\text{kg}\), and \(L=5.6\times10^{-10}~\text{m}\):
\[ \Delta E \approx 9.61\times10^{-19}~\text{J} \approx 6.00~\text{eV}. \]
Step 5: Predict the absorption wavelength
Approximate the electronic transition energy as the photon energy: \( \Delta E \approx \frac{hc}{\lambda} \). Therefore
\[ \lambda \approx \frac{hc}{\Delta E}. \]
With \(c=3.00\times10^8~\text{m/s}\) and the \( \Delta E \) above:
\[ \lambda \approx 2.07\times10^{-7}~\text{m} = 207~\text{nm}. \]
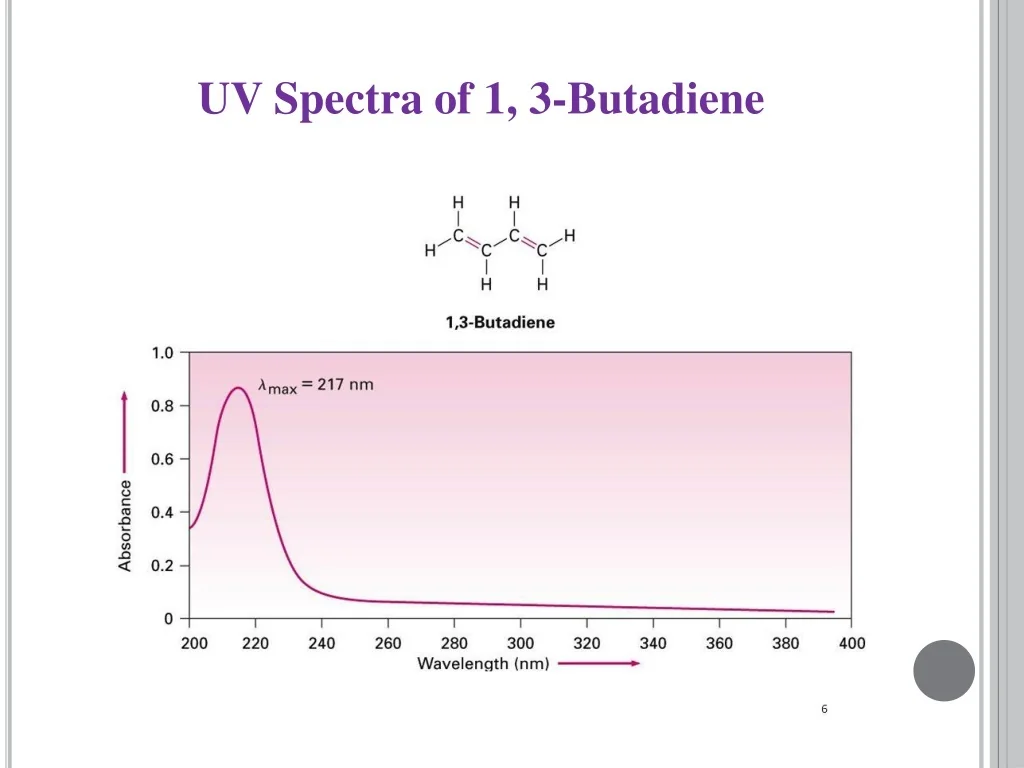
Prediction: butadiene should absorb in the UV (around \( \sim 200~\text{nm} \)), and longer polyenes (larger \(L\)) will shift to longer wavelengths.
Here is an image showing the UV/VIS absorption spectrum of 1,3-butadiene.